
Which substance is being reduced in the equation: Mg + 2HCI -MgCla+H: (a) Mg (b) HCI (c) MgCl (d) Hz 19. What is the oxidation number of As in NaAsO7(a) (b)2 (d) 5 18. Which of the following has the highest entropy Water Ice Water solution or Water vapor water vapor. 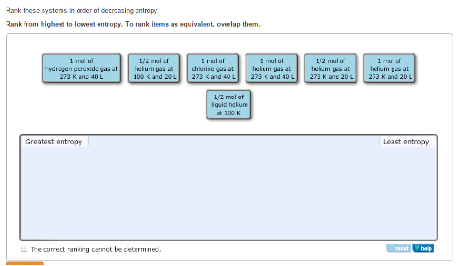
What is the oxidation number of Co in Coci, (a) -3 (b)-6 (c) +8 (d) +3 17. Which one of following systems has the highest entropy Steam at 105 c. Which of the following is not zero for an element in its standard state at 25☌?() AG (b)s (c)AH 16. The value of the equilibrium constant for a particular reaction is 0.48 at 25☌. Which one of the following statements is true about the equilibrium constant for a reaction if AG for the reaction is negative? (a) K-0 (b) K-1 (c)K>1 (d) Kthis dissolving process (a) AH is negative and AS is negative (c) A His negative and AS is positve (b) AH is positive and AS is positive (d) A H is positive and AS is negative 5. Dissolving ammonium chloride in water lowers the temperature of the water. 
Which equation represents a reaction that is decreasing in entropy as the reaction proceeded (a) CaCO3(s) - CO(s) + CO.(B) (c) 2Na(s) + 2H:0(1) - 2NaOH(aq) +H:[g) (b) 2C(s) + O2(g) - 200(g) (d) 2H3(g) + O2(g) + 2H:O(1) 4. In which of the following processes does entropy decrease for the system? (a) boiling water to form steam (c) mixing of two gases into one container (b) dissolution of solid KCl in water (d) freering water to form ice 3. Which one of the following substances has the highest absolute entropy at 25☌? (a) Nib() (b) Neg) (c) H:01) (d) C(s) 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
